From commitment to capability: Are current R&D trends delivering on Africa’s health sovereignty ambitions?
By Impact Global Health and PATH 29 April 2026
Key messages
- Research and development (R&D) investment for Africa’s priority diseasesa is highly concentrated, shaped by epidemic risk and market incentives, rather than current disease burden. This leaves many priority diseases underfunded.
- Clinical research activity in Africa remains uneven, with many priority diseases lacking African-led trials. This limits the continent’s ability to drive its own R&D agenda.
- Africa’s manufacturing capacity is outpacing its clinical research, and until upstream R&D gaps are addressed, health sovereignty will remain a political ambition rather than an operational reality.
a Africa CDC priority epidemic-prone diseases (identified through risk-ranking exercises and listed here alphabetically): chikungunya, cholera, coronavirus diseases (including COVID-19), Crimean-Congo hemorrhagic fever, dengue, Ebola virus disease, influenza (pandemic potential, including avian influenza), Lassa fever, mpox, plague, rabies and Rift Valley fever.
The vision: Africa’s health sovereignty agenda
African leaders have outlined an ambitious vision to strengthen the continent’s health security and reduce reliance on external systems through initiatives such as the Africa Centres for Disease Control and Prevention’s (Africa CDC) Africa Health Security and Sovereignty Agenda and the Accra Initiative. Together, these frameworks emphasize a shift toward self-reliance, focusing on strengthening capacity to prevent, detect, and respond to health threats; expanding local manufacturing of medical countermeasures; and increasing sustainable domestic financing for health innovation. This vision has been further reinforced through commitments made at recent African Union Summits. At the latest summit in February 2026, leaders launched ACHIEVE Africa and endorsed the Presidential Declaration on Advancing Local Manufacturing of Health Products, signaling a continued and accelerated focus on building Africa’s end-to-end R&D ecosystem.
Realizing these ambitions will depend not only on strengthening institutions and manufacturing capacity but also on ensuring that R&D investments are strategically aligned with Africa’s health priorities. Funding patterns play a decisive role in shaping which technologies are developed, which innovation pipelines advance to maturity, and how well health systems are positioned to respond to future threats. Examining how global R&D investments and pipelines align with Africa CDC’s priority diseases provides a critical lens for assessing whether current patterns support or constrain the continent’s health sovereignty ambitions.
Funding trends
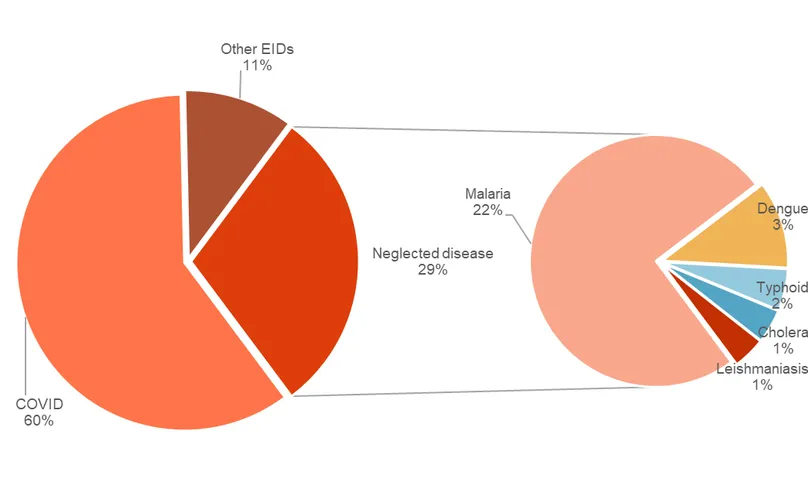
In 2024, the latest year with available data, nearly 60 percent of the US$3.3 billion in global R&D funding for Africa CDC priority diseases was directed to COVID-19. The five priority neglected diseases included in the G-FINDER surveyb1 received 30 percent of the total funding, with malaria alone accounting for three-quarters of that amount, well ahead of dengue (11 percent), the next highest-funded disease. Cholera, typhoid, and leishmaniasis each received only around 5 percent ($40–$50 million) in 2024 (see Figure 1.)
Figure 1. R&D funding for priority diseases by global health area, 2024.
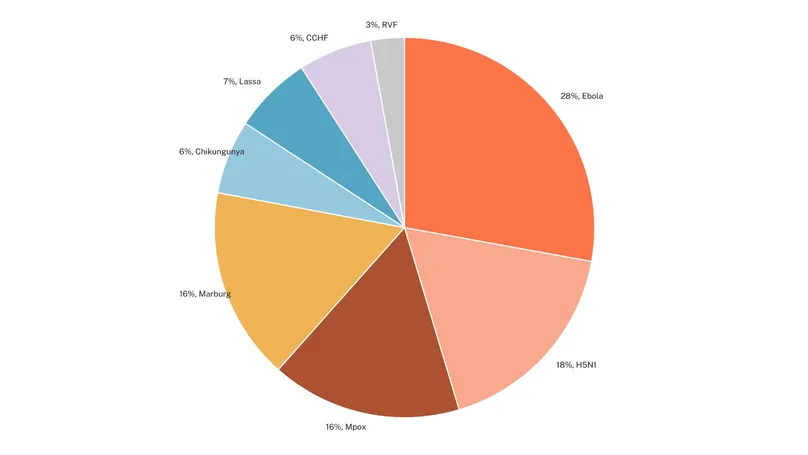
For priority emerging infectious diseases tracked in the G-FINDER survey, 2024 R&D investments were more evenly distributed following post-epidemic declines in COVID-19 funding, ranging from 28 percent ($98 million) for Ebola to 3 percent ($10 million) for Rift Valley fever (RVF) (see Figure 2).
Figure 2. R&D funding for Africa CDC priority non-COVID-19 emerging infectious diseases, 2024.
Taken together, these figures show that R&D investment targeting Africa CDC priority diseases remains highly concentrated in a small number of pathogens. While this pattern may reflect scientific opportunity, epidemic risk, or existing pipeline momentum, it also highlights the challenge of ensuring that investment patterns reflect the full spectrum of Africa’s priority health threats.
A useful way to interpret these funding patterns is to compare them with disease burden in Africa. This relationship is not always straightforward. For emerging infectious diseases, R&D investment is often driven more by future epidemic or pandemic risk than by the current burden of disease. Even for endemic diseases, higher levels of investment may reflect stronger scientific opportunities, while lower levels may result from scientific and development challenges or limited feasibility rather than simply neglect by funders. Still, looking at R&D investment alongside disease burden over time helps show how innovation priorities are aligning with Africa’s health R&D needs.
R&D funding for Africa CDC priority diseases captured in the G-FINDER survey has fallen sharply over the past five years, declining by nearly $1.5 billion per year (14 percent). This drop has been driven primarily by steep reductions in COVID-19 funding in the post-pandemic period, reflectingboth the wide range of countermeasures now available and the roughly 80 percent decline in disease burden in Africa since 2020 (see Figure 3). A similar pattern is evident for Ebola, where falling burden and a relatively mature product landscape have also been accompanied by a marked reduction in R&D funding.
Excluding COVID-19 and Ebola, funding for other priority diseases for which the Institute for Health Metrics’ Global Burden of Disease study data2 are available has remained broadly stable and generally moves in line with disease burden. Dengue stands out, with funding increasing by nearly $7 million (9 percent) per year alongside a 4 percent annual rise in burden. Cholera is the main exception, with funding rising slightly (2.2 percent per year) despite a comparable decline in burden. Overall, across diseases with both burden and funding data points, R&D investment closely tracks disease burden, with a correlation of nearly 90 percent. This suggests that the global innovation system does, in broad terms, respond to changing epidemiological trends over time, but not always quickly or evenly, leaving some high-burden diseases underfunded and slowing the development of essential medical countermeasures.
b The scope of diseases included in this analysis is based on the Africa CDC’s Risk Ranking and Prioritization of Epidemic-Prone Diseases framework published in July 2022, focusing on those for which data are available. This includes key epidemic-prone diseases such as COVID-19, Ebola, Marburg, cholera, chikungunya, Lassa fever, Crimean-Congo hemorrhagic fever, Rift Valley fever, influenza A/H5N1, and mpox, plus endemic diseases in Africa: malaria, leishmaniasis, and typhoid fever.
R&D investment data were obtained through Impact Global Health’s G-FINDER survey, which collects disbursement data from public, private, and philanthropic organizations involved in global health R&D. All grants were verified through a combination of automated and manual quality control processes. The analysis covers funding by global health area and disease, year-on-year funding trends, and comparisons with disease burden using Institute for Health Metrics’ Global Burden of Disease study data. The G-FINDER survey does not collect funding data for plague, anthrax, or rabies.
The foundational clinical research data for this analysis were sourced from the Clinical Trial Community (CTC) platform hosted by nuvoteQ and Science for Africa Foundation (SFA) and subsequently analyzed by Impact Global Health. The pipeline analysis focuses on clinical trials conducted in Africa for the priority diseases, with additional details available via the dashboard on the Impact Global Health website. Further information on the broader R&D pipeline can be found in Impact Global Health’s Infectious Disease R&D Tracker, developed under the Evidence for Impact project. The methodology for pipeline curation and mapping of active clinical candidates across diseases and product areas is oultlined on the website.
Clinical trial capacity in Africa
Clinical trials in Africa have been largely driven by outbreaks, heavily concentrated within five countries, mostly led by non-African institutions, and often misaligned with the disease burden.
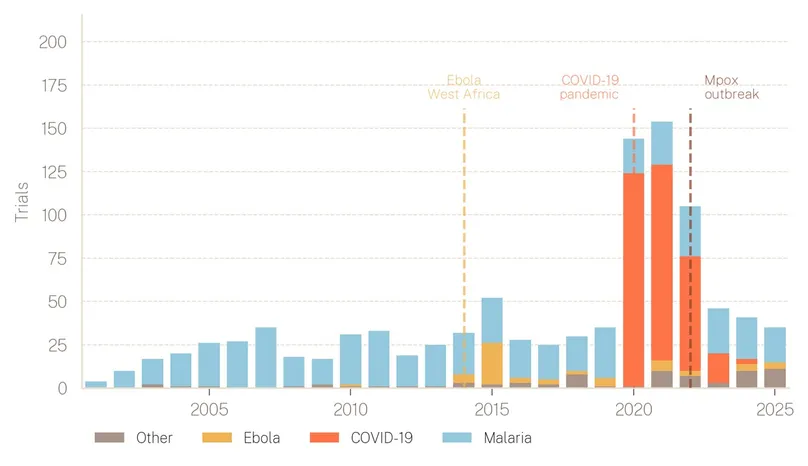
Outbreak drivenc. As Figure 4 illustrates, the 1,008 clinical trials targeting Africa CDC priority diseases from 2001 to 2025 were shaped more by health emergencies than by steady long-term investment. Activity surged during the 2014 West African Ebola epidemic, peaked at 154 trials in 2021 as COVID-19 came to dominate the pipeline, and then subsided as the acute phase passed. From 2001 to 2025, malaria accounted for 55 percent of all trials, and COVID-19 for a further 32 percent, meaning the two diseases together comprised 87 percent of the total. The remaining 11 diseases shared just 13 percent of trials between them.
Figure 3: Five-year trend in disease burden and R&D funding (2019–2024).
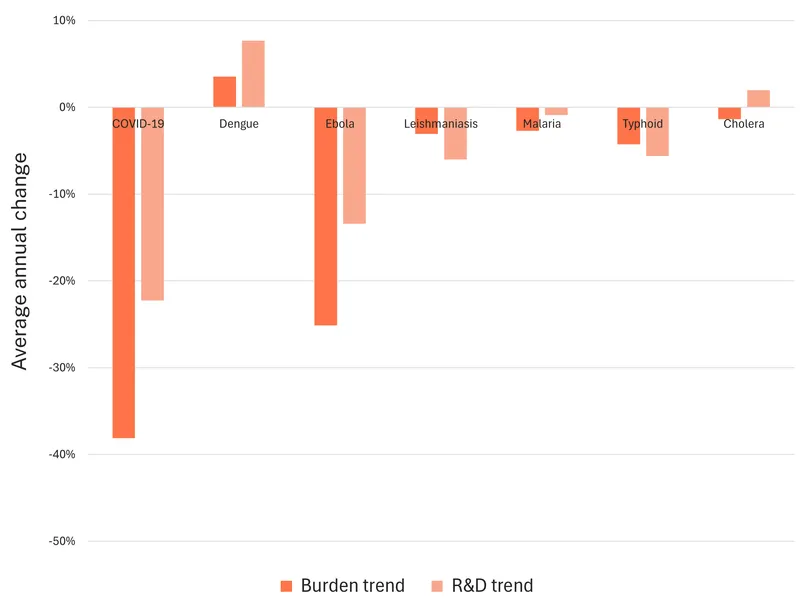
Figure 4: Number of clinical trials for priority diseases, 2001–2025.
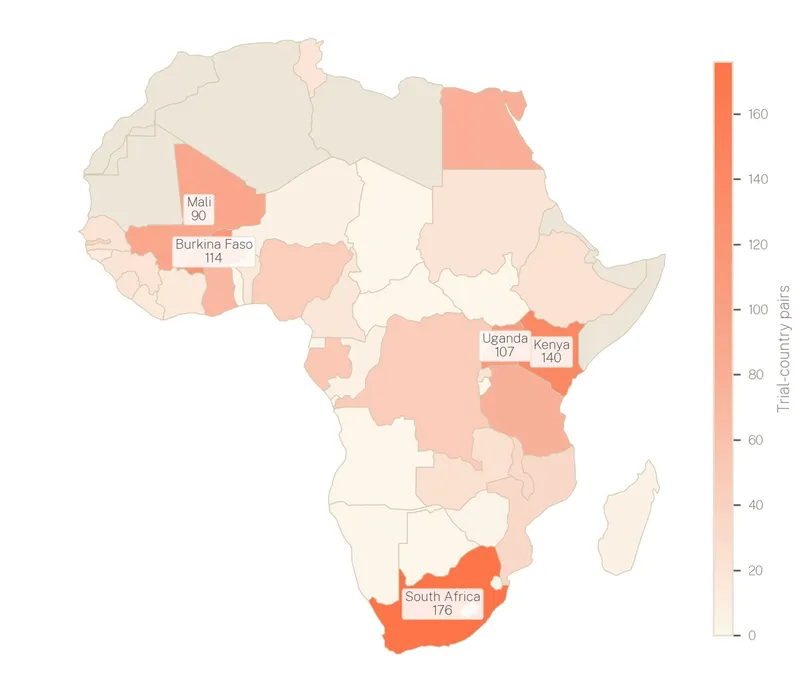
A concentrated geography. Clinical trial activity for the priority diseases spans 41 African countries but is heavily concentrated in a small number of settings, as shown in Figure 5. The top five countries, South Africa (176), Kenya (140), Burkina Faso (114), Uganda (107), and Mali (90), account for most trial-country placements, with South Africa and Kenya alone hosting more trials than the next three combined. West Africa, particularly Burkina Faso and Mali, features prominently, largely because of malaria vaccine trials. As a result, much of the continent remains a blank spot on the research map.
Figure 5. Geographical location of clinical trials in Africa, 2001–2025.d
Research led from elsewhere. Between 2001 and 2025, nearly three-quarters of all clinical trials in Africa were led by institutions headquartered outside the continent (see Figure 6). Of this, non-African universities led 29 percent of trials, global pharmaceutical companies led 26 percent, and government agencies led 14 percent, largely from the United States. African universities accounted for 21 percent, with activity concentrated heavily in Egypt and South Africa. Clinical research capacity in Africa remains tethered to external funding, external protocol design, and external institutional oversight. Building sovereign research infrastructure, where African scientists set the agenda, not merely host the trials, is the central challenge of the next decade.
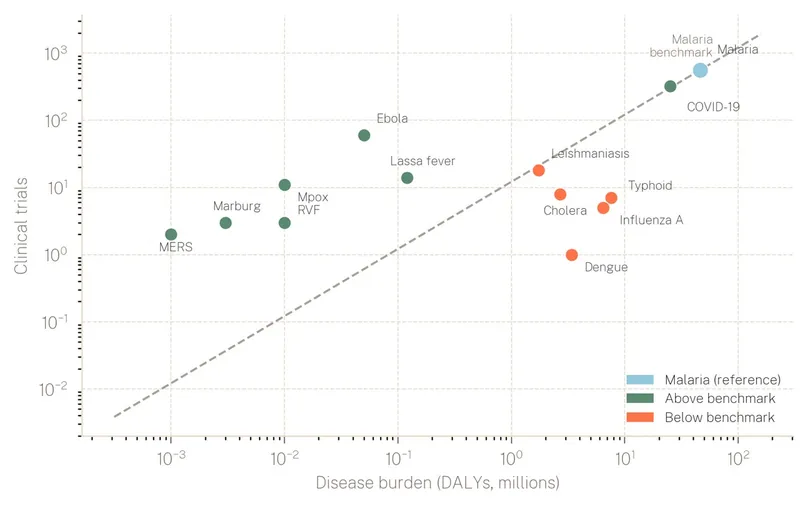
Burden without research. Using malaria, the best-resourced priority disease, as a benchmark for the relationship between cumulative disease burden and R&D investment reveals a substantial misalignment across the landscape (see Figure 7). Several diseases fall well below this benchmark. Cholera, typhoid, and dengue each impose substantial disability-adjusted life year (DALY) burdens yet attract a fraction of the trial activity their toll would warrant. Influenza A (H5N1) follows a similar pattern. By contrast, diseases above the benchmark, Ebola, Lassa fever, mpox, and Marburg, have received disproportionate investment relative to their burden, reflecting the crisis-response dynamic clearly visible in the distribution of trial activity. Addressing this imbalance demands deliberate, sustained investment in the diseases that impose a significant disease burden but lack the political salience to attract it.
Figure 6: Number of clinical trials in Africa by developer/sponsor, 2001 - 2025.
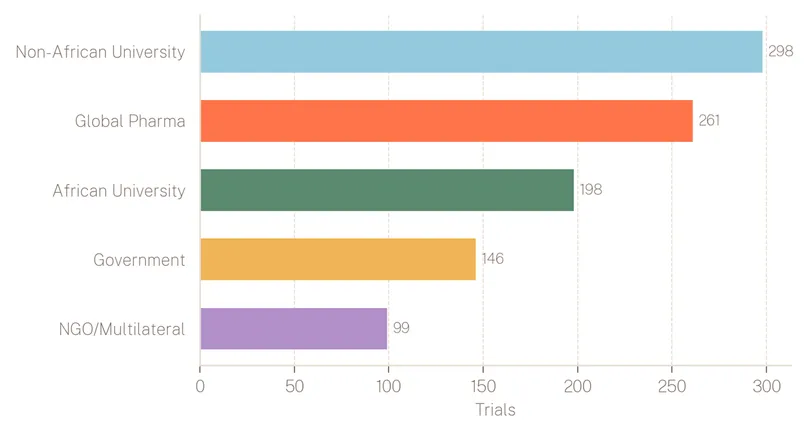
Figure 7: Number of clinical trials by disease burden, 2001–2025.
c Detailed insights into the clinical trials landscape can be accessed through the dashboard provided at https://africa-clinical-trials-capacity.netlify.app
d Trial-country pairs count each combination of a clinical trial and the country in which it is conducted.
Feasibility of local manufacturing in Africa
These findings raise important questions about the feasibility of expanding local manufacturing in Africa. Sustainable manufacturing depends on upstream research, a viable R&D pipeline, and clear pathways from innovation to production. If few products progress through clinical development to approval, African manufacturers will have fewer technologies to license and produce.
Even where research is conducted in Africa, manufacturing pathways often remain unclear. Post COVID-19, Africa has stepped up efforts to expand local manufacturing of medicines, vaccines, and diagnostics. The African Union’s (AU) target to locally manufacture 60% of vaccine needs by 2040 alongside PAVM Partnerships for African Vaccine Manufacturing (PAVM), the AU Roadmap to 2030 & Beyond, and the recent Presidential Declaration on Advancing Local Manufacturing of Health Products in Africa reflect this ambition. But progress has focused mainly on downstream vaccine production, especially fill and finish, with much less attention to upstream capabilities such as product development, antigen production, and process optimization.
Diagnostics have received even less investment. Limited investment, insufficient workforce, and deterring regulations also constrain the competitiveness of diagnostics R&D. According to a diagnostics market failures report by PATH,4 diagnostic companies in Africa that are regionally headquartered have a greater focus on endemic diseases than global companies. This reinforces the need for Africa to shape its own R&D agenda.
Manufacturing feasibility depends on whether key building blocks are in place: availability of technologies to manufacture, technology transfer, regulatory pathways, clinical data, and market demand. These foundations remain uneven. The launch of the African Medicines Agency (AMA) is an important step, but many national regulatory authorities (NRAs) still lack capacity; out of 55 AMA member countries, only 9 have NRAs that have achieved World Health Organization Maturity Level 3.5 This means that most NRAs are still developing and may present unpredictable reviews and approvals, which may discourage investment in early-stage R&D and the introduction of products into the market. Demand signals are also weak, with continued reliance on donor-funded purchasing, limited procurement commitments to locally manufactured products, and low domestic spending on health R&D.3
Investment in Africa also remains concentrated in vaccines and a small number of diseases. Much of this funding is donor driven. Without stronger domestic financing and greater African influence over R&D and manufacturing priorities strategies, the continent’s broader health sovereignty ambitions will be difficult to sustain.
What this means for African health sovereignty
The evidence presented in this brief points to a significant gap between Africa’s health sovereignty ambitions and the R&D foundations needed to realize them.
The continent faces the following challenges:
- Uneven funding. R&D investment targeting Africa’s priority diseases remains highly concentrated toward a small number of pathogens. Epidemic risk and market incentives, not Africa’s public health priorities, continue to shape what gets funded.
- Clustered clinical trials. Clinical trials are concentrated, both in terms of diseases and location. More troubling still, most trials conducted in Africa are led by institutions headquartered outside the continent, largely reducing the continent’s agency.
- Fragmented manufacturing chain. Despite ambitious manufacturing targets, upstream capabilities in product development, antigen production, and diagnostics remain largely underdeveloped. Establishing a sustainable manufacturing ecosystem will require a holistic view of the innovation-to-production pathway and a commitment to upstream research.
- Weak continental coordination. Africa’s R&D and manufacturing ecosystem remains fragmented, with limited coordination across countries on how capabilities should be distributed and developed. Without deliberate regional specialization, countries risk pursuing isolated and duplicative strategies across research, clinical development, and manufacturing, weakening the efficiency, scale, and resilience needed for a functioning continental ecosystem.
No single country can resolve these challenges alone. The research gaps are too broad, the regulatory challenges too complex, and the investment requirements too large for individual national strategies to address them in isolation. Africa’s path to a functioning R&D and manufacturing ecosystem depends on regional specialization and interdependency. Countries must deliberately build complementary capabilities across the value chain, with some specializing in clinical trials and others in product development or manufacturing, coordinated through frameworks such as the AMA and the Regional Economic Communities. Underpinning all of this must be sustained domestic financing mechanisms.
The central question is not whether Africa can produce health technologies. It is whether Africa can shape what gets developed in the first place. Closing the gap between commitment and capability requires influence upstream—over which diseases are prioritized for research, where clinical trials are conducted, and who has ownership of the resulting technologies. Better coordination between African governments, regional institutions, and global funders will be critical to ensuring that future innovation pipelines respond to the continent’s most pressing health threats. The infrastructure being built today is necessary but not sufficient. It must be matched by the products, pipelines, and intellectual ownership that make sovereignty operational rather than aspirational.
Policy recommendations
- Scale domestic investment in health R&D as a driver of economic growth and resilience. African governments should prioritize dedicated financing for health R&D within national budgets and adopt innovative financing models to complement domestic resources. This is not only necessary to ensure research priorities align with national disease burdens and development goals but also to strengthen economic productivity, reduce long-term fiscal pressures, and build the strategic autonomy that health sovereignty demands.
- Ensure external investments build long-term institutional capacity. External health R&D financing has played an important role, but investment partners must move beyond project-based funding. The focus should shift toward helping African institutions define, lead, and sustain their own R&D agendas, rather than hosting work designed and owned elsewhere.
- Fast-track the full operationalization of the African Medicines Agency. A fully functional AMA is critical to converting R&D investments into tangible health outcomes. It will reduce regulatory fragmentation across the continent, accelerate the time to market for African-developed innovations, and strengthen Africa’s readiness to respond to both current and emerging health threats.
- Regional Economic Communities should prioritize coordinated clinical trial execution. Building on progress in regulatory harmonization and manufacturing, Regional Economic Communities should ensure that multi-country clinical trials are coordinated and distributed across member states rather than concentrated in a small number of research-active countries. This will strengthen Africa’s collective ability to generate locally relevant clinical data at the scale the continent’s health challenges require.
- Prioritize collaborative regional models for R&D and manufacturing. Beyond national strategies, African governments should build interdependent regional ecosystems in which countries deliberately specialize across different parts of the value chain, spanning discovery; preclinical and clinical evaluation; chemistry, manufacturing, and control and process development; and manufacturing. Supported by regulatory harmonization frameworks, this approach will enable Africa to develop the complementary capabilities no single country can build alone.
References
1 Africa CDC priority epidemic-prone diseases (identified through risk-ranking exercises and listed here alphabetically): chikungunya, cholera, coronavirus diseases (including COVID-19), Crimean-Congo hemorrhagic fever, dengue, Ebola virus disease, influenza (pandemic potential, including avian influenza), Lassa fever, mpox, plague, rabies and Rift Valley fever.
2 Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease 2023: Findings from the GBD 2023 Study. IHME; 2025. https://www.healthdata.org/research-analysis/library/global-burden-disease-2023-findings-gbd-2023-study
3 Africa Centres for Disease Control and Prevention, Clinton Health Access Initiative, PATH. African Vaccine Manufacturing Mapping, Supply and Demand Landscape. Presented at: Developing Countries Vaccine Manufacturers Network International Annual General Meeting; October 2024; São Paulo, Brazil. https://africacdc.org/download/african-vaccine-manufacturing-mapping-supply-and-demand-landscape/#
4 PATH. Market Failures and Opportunities for Increasing Access to Diagnostics in Low- and Middle-Income Countries. Seattle: PATH; 2022. https://www.path.org/our-impact/resources/market-failures-and-opportunities-for-increasing-access-to-diagnostics-in-low-and-middle-income-countries/
5 Guzman J, Petri-Hidalgo A. A Road Map for Strengthening and Diversifying Regulatory Pathways in Africa. Center for Global Development Policy Paper 377. Washington, DC: Center for Global Development. https://www.cgdev.org/publication/road-map-strengthening-and-diversifying-regulatory-pathways-africa
6 Africa Centres for Disease Control and Prevention. Risk Ranking and Prioritization of Epidemic-Prone Diseases. Addis Ababa: Africa CDC; 2023. Accessed April 22 2026. https://africacdc.org/download/risk-ranking-and-prioritization-of-epidemic-prone-diseases/